 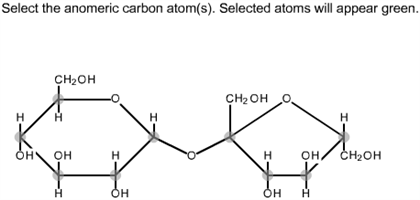
Arrangement of the functional groups can be thus be clear from this conformation of galactose. In this structure, the thicker lines depict the bonds that are closer to the viewer. Galactose is drawn in cyclic structure which is referred as its Haworth projection. Galactose is an example of monosaccharides. This can be generally named as alpha-fructofuranose and beta-fructofuranose. In this way, the anomers of fructose can be identified. If the hydroxyl group in this anomeric carbon is in the axial position, it is said to be alpha fructose and if the hydroxyl group is in the equatorial position, it is said to be beta fructose. 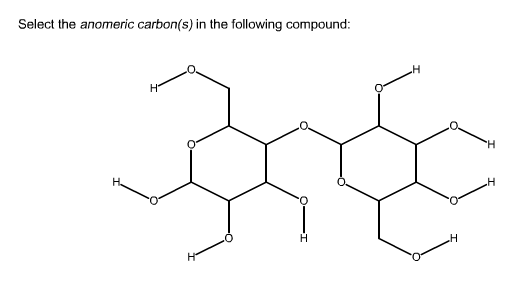
In chair form, the anomeric carbon is the carbon present on the right side of the oxygen atom in the pentagonal ring. This is also applicable to the chair conformation of fructose. In this molecule, if this hydroxyl group is placed below the ring, the molecule is alpha fructose and if this hydroxyl group is attached above the ring, it is a beta fructose. In the pentagonal ring of fructose, the carbon which is placed in the right side of the oxygen is considered as the anomeric carbon. The alpha and beta fructose can be identified from the orientation of the hydroxyl group in the anomeric carbon. As the carbonyl carbon is in the second position, this is the anomeric carbon. It can be drawn in the Haworth projection. In this way, the anomers of glucose can be differentiated.įructose is another monosaccharide with a ketone group in the molecule. If the hydroxyl group is in the equatorial position, it is said to be beta glucose. 
If the hydroxyl group in this anomeric carbon is in the axial position, it is said to be alpha glucose. In chair form, the anomeric carbon is the carbon attached in the right side of the oxygen atom in the hexagonal ring. This is also applicable to the chair conformation of glucose. On the other hand, if this hydroxyl group is attached above the ring, it is a beta glucose. If the hydroxyl group attached to the anomeric carbon is placed below the ring, the molecule is alpha glucose. In the hexagonal ring of glucose, the carbon atom placed in the right side of the oxygen, is the anomeric carbon. From the position of the hydroxyl groups present in the ring its alpha and beta form can be differentiated. The anomers of glucose can be understood from this conformation. Arrangement of the functional groups can thus be clear from this conformation of glucose. In this structure, the thicker lines depict the bonds which are closer to the viewer. Glucose is drawn in cyclic structure which is referred as its Haworth projection. The main storage polysaccharide of animal cellsĪ fibrous, tough, water-insoluble substance found in the cell walls of plants (esp.Glucose is an example of monosaccharides. Heteropolysaccharides = contain two or more different kinds Homopolysaccharides = contain only a single monomeric species What is the difference between homopolysaccharides & heteropolysaccharides? identity of their recurring monosaccharide units The end of a chain in a disaccharide or polysaccharide with a free anomeric carbonĤ ways glycans (polysaccharides) differ from each otherġ. Joins the anomeric carbon of a sugar to a nitrogen atom in glycoproteins & nucleotides When a hydroxyl group of one sugar molecule (cyclic) reacts with the anomeric carbon of the other covalently joins two monosaccharides to form a disaccharide Sugars capable of reducing cupric ion ex) glucose When one ring form opens briefly into the linear form, then closes again to produce the other anomer ex) the alpha & beta anomers of D-glucose interconvert in aqueous solution Haworth perspective formulas (NOT Fischer projections used for linear sugar structures) What type of representation is best for cyclic sugar structures? Six-membered ring compounds named so because they resemble the six-membered ring compound "pyran" What is the relationship between anomers & anomeric carbon?Īnomers = isomeric forms of monosaccharides that differ only in their configuration about the hemiacetal or hemiketal carbon atomĪnomeric carbon = the carbonyl carbon atom Hemiketal = the derivative of a reaction between alcohols & ketones Hemiacetal = the derivative of a reaction between alcohols & aldehydes What is the difference between a hemiacetal & a hemiketal? Two sugars that differ only in the configuration around one carbon atom ex) D-glucose & G-mannose differ only in stereochemistry at C-2, D-glucose & D-galactose Three-dimensional sugar structures on paper What do Fischer projection formulas represent? Ketose = a monosaccharide where the carbonyl group is at any other position What is the difference between a ketose & an aldose?Īldose = a monosaccharide where the carbonyl group is at an end of the carbon chain 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
